Orthopedic Surgeon Email List: How Medical Device and Implant Companies Reach Surgical Specialists

Tom Reardon had spent twelve years in spine device sales before he moved into a commercial operations director role at a mid-sized orthopedic implant company. His first assignment was clear: build the outreach infrastructure for a new lumbar fusion system launch. The clinical data was strong. The technique was differentiated. But the problem was reach.
His field team of 28 reps covered roughly 600 surgeons through existing relationships. That was a solid base. But it was a fraction of the addressable market. The company’s marketing team had been relying on conference badge scans and event lists from the past three years. Combined, those sources gave them about 1,100 orthopedic contacts nationally. But when they ran the list against their CRM, 34% of the emails were invalid, duplicated, or belonged to contacts who had changed institutions.
What Tom needed was a verified orthopedic surgeon email list built from current NPI data. It had to be filterable by subspecialty and procedure focus. So his team could prioritize the surgeons most likely to adopt a new lumbar fusion technique. This guide covers what he needed to know. It also covers what every medical device commercial team should understand about reaching orthopedic surgeons through email in 2026.
What Is an Orthopedic Surgeon Email List?
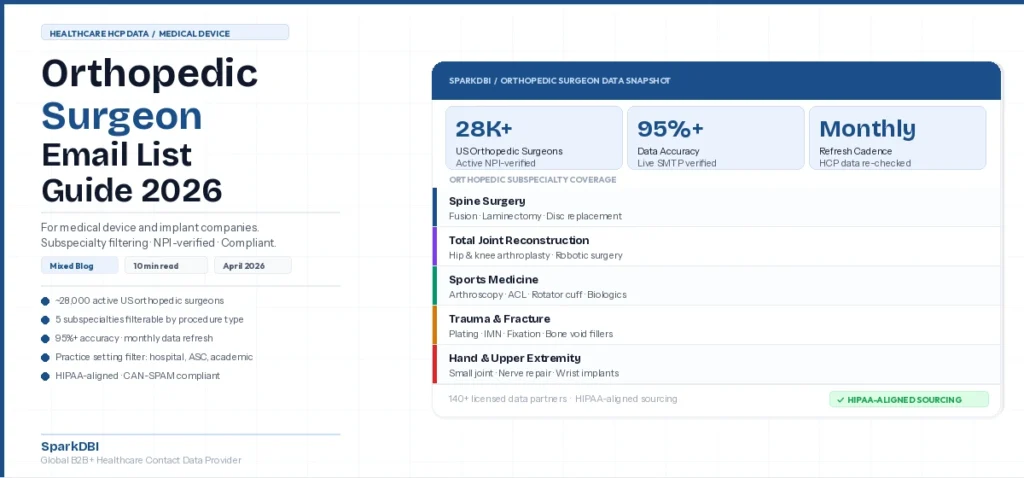
An orthopedic surgeon email list is a verified database of contact information for licensed orthopedic surgeons. It includes confirmed professional email addresses, NPI numbers, subspecialty classifications, procedure focus areas, practice settings, and geographic identifiers. Quality orthopedic surgeon email lists draw from NPI registry cross-matching, medical board licensing data, and live email deliverability verification — not scraped conference registrations or unverified purchased files.
SparkDBI maintains NPI-verified orthopedic surgeon contact data as part of its healthcare professional database of 10,380,376 verified HCP contacts spanning 39 specialty groups. The data is filterable by subspecialty, procedure type, practice setting, and state. The orthopedic segment supports targeting by sub-discipline. That means spine surgery, sports medicine, and joint reconstruction are all separable — which matters for device campaigns where indication aligns with specific procedures, not orthopedics broadly.
Why Orthopedic Surgeons Are the Primary Target for Medical Device Companies
Orthopedic surgery drives one of the highest-volume medical device purchasing categories in the US. Joint replacement hardware, spinal implant systems, arthroscopic instruments, bone fixation devices, and sports medicine biologics all flow through orthopedic surgeons. These surgeons influence or directly determine procurement decisions at their institutions and practice groups.
According to the American Medical Association’s Physician Workforce Data, there are approximately 28,000 active orthopedic surgeons in the United States. But the distribution within that population is highly uneven for commercial purposes. The 3,000 to 5,000 high-volume fellowship-trained subspecialists account for a disproportionate share of device adoption decisions. This group includes spine surgeons, total joint reconstruction surgeons, and sports medicine surgeons at high-volume centers.
Because of this, reaching orthopedic surgeons at scale requires subspecialty filtering. A campaign that treats all 28,000 orthopedic surgeons as a single audience will waste most of its reach budget. Most recipients will have no relevant indication for the product.
Orthopedic Subspecialties: How to Segment for Device Campaigns
SparkDBI’s orthopedic surgeon data supports subspecialty filtering across the major surgical practice areas relevant to medical device commercial programs. Here is how each segment maps to device categories, and why each one requires separate targeting.
Spine Surgeons
Spine surgeons perform cervical, thoracic, and lumbar procedures including fusion, laminectomy, disc replacement, and deformity correction. They are the primary target for spinal implant systems, interbody cages, pedicle screw constructs, and minimally invasive access systems. Spine surgery spans both orthopedic and neurosurgery training pathways. So reaching verified spine specialists requires specialty cross-referencing, not a simple orthopedic filter.
Total Joint Reconstruction Surgeons
Total joint surgeons focus on hip and knee arthroplasty — the highest-volume elective orthopedic procedure category in the US. They drive purchasing decisions on implant systems, bearing surfaces, fixation interfaces, and robotic surgery platform adoption. Fellowship-trained arthroplasty surgeons at high-volume centers have disproportionate influence on health system formulary decisions. Each surgeon may perform hundreds of cases per year — which means their adoption decisions carry significant procurement weight.
Sports Medicine and Arthroscopic Surgeons
Sports medicine orthopedic surgeons perform shoulder, knee, and ankle arthroscopy — including ACL reconstruction, rotator cuff repair, labral procedures, and cartilage restoration. This group drives adoption of anchors, suture systems, biologics like PRP and stem cell constructs, and next-generation graft materials. However, they tend to operate in ambulatory surgery center settings, which carry different procurement dynamics than hospital-based practice.
Trauma and Fracture Surgeons
Orthopedic trauma surgeons specialize in the surgical management of fractures, dislocations, and skeletal injuries. They are the primary target for plating systems, intramedullary nailing, external fixation constructs, and bone void fillers. Trauma surgeons typically practice in academic medical centers, Level I and Level II trauma centers, and large regional hospital systems. In those settings, device formulary decisions involve department committees and supply chain contracting teams.
Hand and Upper Extremity Surgeons
Hand surgeons manage fractures, tendon repairs, nerve reconstruction, and small joint arthroplasty of the wrist, hand, and elbow. This group is the target for microsurgical instruments, small bone fixation systems, wrist and hand implants, and nerve repair biologics. Hand surgery is a fellowship subspecialty with its own distinct procedure mix. So these surgeons need separate targeting from general orthopedic campaigns.
Filter by Orthopedic Subspecialty and Procedure Focus
SparkDBI’s healthcare email lists by specialty let you filter orthopedic surgeon contacts by subspecialty, practice setting, geographic market, and institution type. See live record counts for your exact target segment before committing to a data package.
Why Conference Lists and Badge Scans Don’t Scale
Tom’s conference list problem is common in medical device commercial teams. Badge scans from AAOS, North American Spine Society, and specialty society meetings feel like high-quality data. After all, these contacts are self-selected clinicians who attended a relevant conference. But as a primary list-building strategy, conference registrations have four structural limitations that make them inadequate for national outreach programs.
Coverage ceiling: even the largest orthopedic meetings attract a fraction of practicing surgeons. For example, AAOS annual meeting registration typically covers 10,000-15,000 attendees. That is a substantial event. But it still represents under 60% of the active orthopedic surgeon population.
Data decay: conference email addresses are often personal or institutional addresses that change with every job transition. A badge scan from three years ago carries the same decay risk as any other unverified list. That means roughly 25-30% invalid addresses per year, regardless of how the data was originally collected.
No subspecialty depth: conference registration data rarely includes procedure-level specialty classification. Without subspecialty depth, you cannot distinguish a spine surgeon from a sports medicine surgeon from a trauma specialist in the same dataset.
Consent and compliance complexity: badge scan consent is typically scoped to the exhibitor who scanned the badge, not to all subsequent marketing communications. So using conference lists for ongoing email programs without proper opt-in management creates CAN-SPAM compliance exposure.
NPI-verified data from a structured HCP data provider solves all four problems. It provides national coverage, monthly email deliverability verification, subspecialty filtering, and compliant data sourcing documentation. That documentation supports enterprise legal and compliance review. As a result, teams get everything they need to move from outreach planning to campaign launch without friction.
Quality vs Commodity: What Separates a Verified Orthopedic Surgeon List
| Data Attribute | SparkDBI NPI-Verified List | Commodity Conference / Scraped List |
|---|---|---|
| NPI Matching | Cross-referenced against CMS registry for active status | Self-reported, no registry confirmation |
| Subspecialty Depth | Spine, total joint, sports medicine, trauma, hand surgery | Broad “orthopedic surgery” only |
| Email Verification | Monthly live SMTP check, catch-all flagged | No ongoing deliverability check |
| National Coverage | All 50 states, all practice settings | Event-biased, geography skewed |
| Hard Bounce Rate | Under 3% in live campaigns | 15-40% after 12 months without re-verification |
| Compliance Docs | HIPAA-aligned sourcing, opt-out management | Badge consent scoped to specific exhibitor |
How SparkDBI Builds and Maintains Orthopedic Surgeon Contact Data
SparkDBI is a global B2B and healthcare contact data provider with 270M+ verified contacts across 200+ countries and 140+ licensed data partners. The orthopedic surgeon dataset sits within SparkDBI’s broader HCP database of 10,380,376 verified contacts spanning 39 specialty groups. It carries monthly refresh and 95%+ accuracy across all specialty segments.
SparkDBI’s team constructs each orthopedic surgeon record through a multi-source verification process:
- CMS NPI registry cross-matching: every orthopedic surgeon record is matched against the National Provider Identifier registry to confirm active license status, specialty taxonomy, and current practice state
- State medical board and specialty board data: supplemented with board certification records to confirm fellowship training and subspecialty credentials where available
- Monthly live email deliverability verification: SparkDBI checks every email address against live SMTP responses on a monthly refresh cycle, treating catch-all institutional domains separately from standard checks
- 140+ licensed data partner inputs: sourced from licensed provider directories, medical association records, and professional networks, with deduplication applied across all sources
- Practice setting and institution classification: records include practice setting flags — hospital, academic medical center, ambulatory surgery center, group practice — so device commercial teams can filter by the procurement environment that matters for their product
You can verify live record counts and last-refresh dates for the orthopedic surgeon segment on SparkDBI’s live healthcare data dashboard before making any purchasing commitment.
See the Orthopedic Surgeon Data Before You Buy
SparkDBI’s HCP email list data cards show verified record counts, field coverage, and refresh dates for the orthopedic surgeon segment and every other HCP specialty. No commitment required to see the full data specification.
Compliance Considerations for Medical Device Email Outreach to Surgeons
Medical device companies emailing orthopedic surgeons operate under several overlapping compliance requirements. Understanding these before building outreach programs prevents the compliance reviews that delay launches. It also prevents campaigns stalling at the worst possible moment.
CAN-SPAM compliance: Commercial emails to surgeons at their professional NPI-registered addresses require a functional opt-out mechanism, a physical mailing address, and an honest subject line. You must honor opt-out requests within 10 business days. No prior consent is required to initiate contact. The FTC’s CAN-SPAM compliance guide outlines these requirements in full.
HIPAA scope: HIPAA’s Privacy Rule covers protected health information that covered entities hold about patients. It does not restrict medical device companies from using professional contact data to reach licensed surgeons in their professional capacity. So emailing an orthopedic surgeon about a fusion system using their professional email address is not a HIPAA matter.
FDA and Sunshine Act considerations: medical device promotional communications are subject to FDA regulations on truthful and non-misleading promotion. The Sunshine Act requires reporting of transfers of value to covered recipients, including physicians. Educational content delivered via email is generally not a reportable transfer of value. However, marketing programs with embedded offers — sample kits, educational dinners, trial programs — may require Sunshine Act reporting. The structure of the program determines whether reporting applies.
SparkDBI sources its orthopedic surgeon data under HIPAA-aligned practices with documented data lineage and opt-out management infrastructure. So enterprise legal and compliance teams get the documentation they need without extra legwork.
What Tom’s Team Achieved
Tom’s team replaced their stale 1,100-contact conference list with NPI-verified orthopedic surgeon data. They segmented it across three subspecialty tracks: spine surgeons for the lumbar fusion system launch, total joint surgeons for an adjacent product line, and sports medicine surgeons for a biologics pilot program. Each track received separate messaging aligned to its procedure focus.
The verified dataset delivered 8,400 orthopedic surgeon records across the three segments. That compared to just 726 usable contacts surviving deduplication of the original conference list. The first launch wave targeted 3,200 spine surgeons in the states where the field team had strongest coverage. Hard bounce rate on that send: 1.6%. Open rate: 31%, compared to the 18% industry average the team had been tracking on prior conference-list campaigns.
Six weeks later, 47 surgeons who had engaged with the email sequence requested a rep visit or clinical evaluation meeting. That was more inbound surgeon interest in six weeks than the conference list had generated in the prior eighteen months.
The data didn’t replace the field team. It told them exactly where to deploy their time.
Five Questions to Ask Before Buying an Orthopedic Surgeon Email List
1. Is every record NPI-matched against the CMS registry? Not just “NPI-sourced.” It must be actively cross-referenced to confirm the surgeon holds an active license and that their specialty taxonomy matches your target.
2. When were email addresses last verified for live deliverability? Acceptable answer: within the past 90 days. If the vendor cannot give a specific date, the data has not been recently verified.
3. Can you filter by orthopedic subspecialty? Spine, total joint, sports medicine, trauma, and hand surgery should be separable segments. Any vendor who can only deliver broad “orthopedic surgery” cannot support subspecialty-targeted device campaigns.
4. Can you filter by practice setting? Academic medical center, community hospital, ambulatory surgery center, and group practice carry very different procurement dynamics. The right contact depends on where your device actually gets used and purchased.
5. Will you provide a sample for independent verification before purchase? Any vendor who refuses to provide 300-500 sample records for independent deliverability testing should not receive your budget.
Frequently Asked Questions
What is an orthopedic surgeon email list?
An orthopedic surgeon email list is a verified database of professional contact information for licensed orthopedic surgeons. It includes confirmed email addresses, NPI numbers, subspecialty classifications, practice settings, and geographic identifiers. Quality lists draw from NPI registry cross-matching and monthly live email deliverability verification — not conference registrations or unverified purchased files.
How many orthopedic surgeons are in the US?
There are approximately 28,000 active orthopedic surgeons in the United States, according to the American Medical Association’s Physician Workforce data. For medical device commercial programs, the high-value target is typically narrower. The 3,000 to 5,000 fellowship-trained subspecialists in spine surgery, total joint reconstruction, and sports medicine drive the majority of device adoption decisions at their institutions.
Can medical device companies legally email orthopedic surgeons?
Yes. US law permits medical device companies to email orthopedic surgeons at their professional NPI-registered addresses for product awareness and promotional campaigns. HIPAA’s Privacy Rule applies to patient health information, not professional outreach to licensed surgeons. All commercial email programs must comply with CAN-SPAM for opt-out management. Device promotional content must also meet FDA regulations on truthful and non-misleading promotion.
Why is subspecialty filtering important for orthopedic surgeon outreach?
Subspecialty filtering is important because orthopedic surgeons practice across fundamentally different procedure categories with different device needs. A spine surgeon has no need for a hip arthroplasty system. A sports medicine surgeon does not perform spinal fusion. Without subspecialty filtering, device campaigns reach a broad audience where most recipients have no indication for the product. That wastes budget and dilutes engagement signals that affect future deliverability.
What bounce rate should I expect from a quality orthopedic surgeon email list?
A properly NPI-verified orthopedic surgeon email list with recent deliverability verification should produce hard bounce rates below 3% in live campaigns. Orthopedic surgeons frequently practice at hospital and academic medical center domains. Many of those domains use catch-all email configurations. Lists without specific catch-all handling tend to produce bounce rates 2-3x higher than properly verified lists. Always ask your data vendor how they handle catch-all domains — not just standard SMTP verification.
How does SparkDBI source orthopedic surgeon contact data?
SparkDBI sources orthopedic surgeon contact data through NPI registry cross-matching, state medical board licensing records, 140+ licensed data partner inputs, and monthly live email deliverability verification. The team confirms every record against the CMS National Provider Identifier registry for active license status and specialty taxonomy. SparkDBI refreshes HCP records monthly — including orthopedic surgeon data. As a result, the platform maintains 95%+ accuracy across all surgical specialty segments.
Key Takeaways
- There are approximately 28,000 active orthopedic surgeons in the US. Subspecialty segmentation by spine, total joint, sports medicine, trauma, and hand surgery is required for effective device campaigns.
- Conference badge scans cannot replace NPI-verified data. They carry coverage ceilings, no subspecialty depth, ongoing decay risk, and consent limitations.
- Practice setting filtering matters as much as subspecialty. Hospital, ASC, and academic medical center contacts carry very different procurement dynamics.
- Medical device email outreach to orthopedic surgeons is CAN-SPAM compliant when executed correctly. No prior consent is required, but opt-out management is mandatory.
- SparkDBI maintains NPI-verified orthopedic surgeon data with monthly refresh, 95%+ accuracy, subspecialty filtering, and practice setting classification across all 50 states.
- Always request a sample for independent verification before purchasing any HCP list. Any vendor who refuses is not worth trusting with your campaign budget.
Get Verified Orthopedic Surgeon Data for Your Device Campaign
SparkDBI provides NPI-verified orthopedic surgeon email lists filterable by subspecialty, practice setting, and geography. 95%+ accuracy, monthly refresh, HIPAA-aligned sourcing. Request 50 free verified contacts before committing. Get 50 Free Surgeon ContactsView Live HCP Dashboard
